Placental Function Testing
- Maternal biochemistry (blood tests in the mother)
- Placental ultrasound
- Uterine artery Doppler (measuring blood flow from the mother to the placenta)
- Umbilical artery Doppler (measuring blood flow from the fetus to the placenta).
Overview of tests
There are several choices of prenatal screening tests available to determine the risk of having a baby with Down syndrome and to screen for spina bifida and other birth defects. Screening tests for Down syndrome combine the results of the analysis of proteins in the mother’s blood with an ultrasound of the fetal neck. These screening tests are described below:
Maternal biochemistry
Maternal biochemistry tests (blood tests in the mother) are conducted at different times throughout pregnancy to detect appropriate levels of proteins in the mother’s blood.
First trimester screen (FTS): The first trimester screen (FTS) is performed at 11-13 weeks of gestation and is comprised of a blood test to measure levels of two proteins in the mother’s blood: 1) pregnancy-associated plasma protein-A (PAPP-A), and 2) human chorionic gonadotropin (hCG). Furthermore, to assess baby’s risk of developing Down syndrome, maternal age and the measurement of fluid at the back of the fetal neck (called nuchal translucency) are also used. This is the test most commonly used at Mount Sinai.
Maternal serum screen (MSS): The maternal serum screen (MSS) blood test is performed at 15-19 weeks of gestation and measures four proteins in the mother’s blood: 1) alpha-fetoprotein (AFP), 2) total human chorionic gonadotropin (total hCG), 3) estriol (uE3), and 4) inhibin (dimeric inhibin assay or DIA).
Integrated Prenatal Screen (IPS): The integrated prenatal screen (IPS) is performed at 15-19 weeks of gestation and combines the elements of the FTS and MSS to provide a more accurate determination of the risk of having a baby with Down syndrome. This test has a lower false-positive rate (i.e. the test falsely predicts the syndrome while the baby is born without it), and includes the risk assessment for spina bifida (measured by levels of AFP).
Reinterpreting serum screen results: It is important to note that these biochemical markers are currently used only in prenatal screening to indicate the risk of a chromosomal abnormality (mainly Down syndrome) and spina bifida. If the screening test results predict a high-risk of Down syndrome, the most common action is to offer a diagnostic test called amniocentesis, which will determine if a chromosomal abnormality exists. Since amniocentesis carries a small risk of pregnancy complications (approximately 1 in 500), an alternative choice is to have an early detailed fetal anatomical ultrasound examination to look for specific markers that suggest Down syndrome (such as the nuchal translucency). The same ultrasound examination is performed in women with elevated AFP to look for evidence of spina bifida. The diagnosis must always be confirmed by an amniocentesis before any action is taken on screening test results.
If women have abnormal results for these biomarkers, the chances are much greater that their fetus is normal (approximately 29 in 30) than it has either Down syndrome or spina bifida (approximately 1 in 30).
Knowing that an amniocentesis test is normal and/or that the fetal anatomical ultrasound (US) is normal is an immense relief for parents, but cannot guarantee that there is no risk in the pregnancy. A common explanation for false-positive results is that the placenta is not working properly and is responsible for the abnormal protein levels in maternal blood, as shown below.
Having more than one abnormal test result is strongly associated with stillbirth or extremely preterm birth (<32 weeks’ gestation) due to pregnancy complications from placental insufficiency.
Placental ultrasound
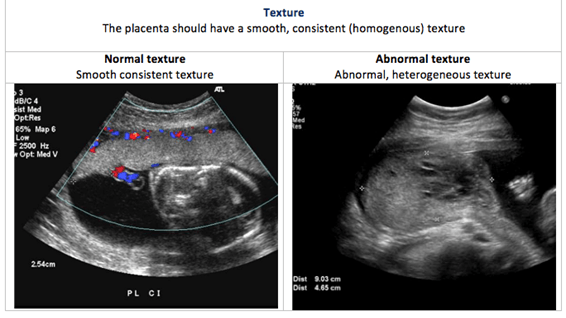
Placental US is performed to determine the size, shape and texture of the placenta. This is called placental morphology US.
US is a non-invasive diagnostic tool to visualize and measure the developing fetus and its surrounding structures (amniotic fluid, umbilical cord, placenta, uterus and cervix). US shows the developing fetus in real time. At present, the ideal window for the placental morphology US is approximately 19-22 weeks (combined with, or near to, the time of routine fetal anatomical US).
Placental size, shape and texture: The placental US assesses the placental morphology, which includes placental size, shape and texture. Each of these is compared between normal and abnormal placentas in the images below:
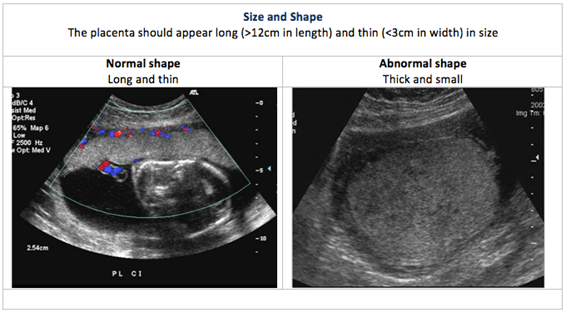
Placental location: In a typical pregnancy, the placenta attaches to the upper portion of the uterus (called the fundus). However, when the placenta is in the lower uterine segment of the uterus, it may cover part of all of the internal os (opening) of the cervix. A low-lying placenta near or covering the internal os is termed placenta praevia. Placental location is part of the fetal anatomical US examination.
Umbilical cord insertion: At the end of the first trimester of pregnancy (12 weeks), the placenta has become distinct from the membranes. Using US, we can visualize the development of the placenta and the location of the umbilical cord. Typically, the umbilical cord is seen to insert in the central part of the placental disc (central cord insertion). If part of the placenta becomes damaged or does not develop properly, then that area of the placenta thins out into membranes, leaving the umbilical cord originating closer to the edge of the placental disc. This could increase the risk of developing vasa praevia during pregnancy.
Number of blood vessels in the umbilical cord: The umbilical cord usually has three blood vessels; two arteries and one vein. The arteries transport deoxygenated blood and waste from the fetus to the placenta. The umbilical vein carries oxygenated blood and nutrients from the placenta to the fetus to support its growth and development. Occasionally, the umbilical cord contains only one umbilical artery (called a two-vessel cord or single umbilical artery). When this occurs, the remaining umbilical artery is wider, and thus has a compensatory larger volume of blood flow. Rarely, this compensation does not occur, and fetal growth may be restricted. Conversely, a single umbilical artery may have nothing to do with abnormal placental development but may be associated with birth defects (for example, a missing kidney) or other more serious problems. Therefore, fetuses with a 2-vessel-cord require a detailed anatomical US examination.
Uterine artery Doppler
Uterine artery Doppler testing measures blood flow from the mother to the placenta via her uterine arteries.
During pregnancy, the uterine arteries enlarge and dilate; this permits a larger volume of blood to flow into the placenta. The maternal blood vessels feeding the placenta also relax to lower resistance, thus increasing the blood flow into the placenta. Doppler images such as the one below, with their dips and peaks, can tell specialists how well the blood is flowing through the uterine blood vessels, and whether there is the right amount of resistance.
Umbilical artery Doppler
Umbilical artery Doppler testing uses the same technology as the uterine artery Doppler to measure the fetal blood flow to the placenta.
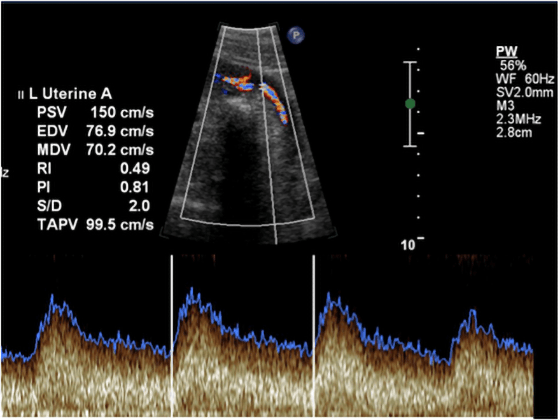
An abnormal uterine artery Doppler could indicate that there is lower blood flow and less nutrient-rich blood for the developing fetus. This could imply not only a restricted blood supply, but is also a significant risk factor for long-term maternal cardiovascular disease. Women with abnormal uterine artery Doppler will be monitored closely, as they may be at risk for preterm delivery (<32 weeks).
Placental Growth Factor (PlGF)
PlGF is produced mainly by the developing placenta (trophoblast) and is secreted into maternal blood. The levels of PlGF rise during the second trimester, such that by the time the fetus is viable, normal levels of PlGF strongly indicate normal placental function.
In women with suspected “placental insufficiency” i.e. with elevated blood pressure or a small for dates uterus, low levels of PlGF (<100 pg/mL) can direct clinicians to do further testing for maternal pre-eclampsia and/or fetal growth restriction (FGR). Approximately 1/3 of such women will be found to have pre-eclampsia and/or FGR. Conversely, if PlGF levels are normal in this context, 98% of women will have healthy pregnancies for the subsequent 2 weeks (negative predictive value = 98%).
The test is valid from 22-36 weeks gestation.
The PlGF blood test result is available in 1-2 hours at Mount Sinai Hospital and therefore is a substantial aid to clinical care. Women with low PlGF levels can have direct access to MFM expertise for advice and care. Mount Sinai is the first hospital in Canada to provide diagnostic PlGF testing in pregnancy for suspected pre-eclampsia or FGR.
